Update (April 2, 2020):
South Korea’s foreign ministry on March 28 said that the U.S. Food and Drug Administration (FDA) gave three South Korean COVID-19 test kit makers obtained preliminary approval. The ministry expects that the pre-approvals would pave the way for detection kits to enter the United States.
The initial FDA approval would enable the Korean kit manufacturers to sell their products in the United States, where the total number of confirmed COVID-19 infection cases already exceeded 100,000.
However, the ministry did not provide the manufacturers’ names nor a time frame for their possible sale in the U.S.
Original:
South Korean diagnostics companies, such as Seegene, KogeneBiotech, and PCL, specializing in test kits for detecting infections, are launching their latest real-time polymerase chain reaction (PCR) COVID-19 assay kits locally and to destinations abroad.
Medical experts believe that the vaccine for the novel coronavirus would take at least providers a year to develop. The most currently realistic preventive measure is to present an immediate diagnosis and follow up with an effective quarantine.
As of Tuesday afternoon, the number of confirmed cases in the country has risen to 977, with a great deal coming from the city of Daegu. Market watchers said that pharmaceutical companies seem to have the test kit situation under control. However, test kits cost around US$132 (160,000 won).
Existing tests are only free to those who have been diagnosed with symptoms by a doctor, those who are showing signs within 14 days of close contact with a confirmed patient, or those who have visited China and present symptoms within 14 days.
Korea Centers for Disease Control and Prevention (KCDC) said that the test involves the collection of nasal discharge and sputum. The samples are induced from the lower respiratory tract, which for most patients are an uncomfortable experience.
KogeneBiotech, PCL, Seegene
Last month, the KCDC released a real-time PCR test in collaboration with KogeneBiotech that can produce results in less than six hours through the amplification of targeted DNA. Previous methods required up to two days for detection.
KogeneBiotech said it used the World Health Organization’s (WHO) guideline in creating real-time PCR technology. The medical tech manufacturer was the first to release its PowerChek 2019 nCoV Real-Time PCR Kit to acquire emergency approval for use and listing from the state agency.
Over 50 hospitals designated to test suspected infected patients in the country are being supplied with KogeneBiotech’s real-time kit. The company said it is also starting to assess the test in countries including China, Japan, Vietnam, United Arab Emirates, Saudi Arabia, Bahrain, and Iraq.
Generally, the Ministry of Food and Drug Safety requires such new test kits a year to pass regulations. However, due to the nature of the situation, the ministry temporarily relaxed the rules to allow for a speedy response.
PCL, a provider of in vitro diagnostic products, partnered with OliX Pharmaceutical in developing a real-time PCR kit. A PCL official said that the company already applied for approval with the KCDC. The official added that they are awaiting licensing so that could begin exporting in China and India.
Meanwhile, Seegene’s AllplexTM 2019-nCov assay kit had obtained marking and emergency use authorization from the KCDC last week. The assay kits also acquired European approval and Seegene currently supplies them domestically, as well as in Italy and Germany.
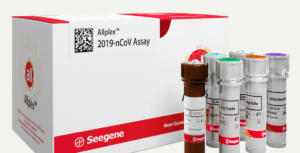
Seegene’s kit detects and identifies the COVID-19 using three target genes. The kit increased output using test in one tube and automation system.







